Third-Party Verified Purity & Identity Testing
Independent laboratory testing is performed on batches to verify compound identity, purity, and composition using HPLC and Mass Spectrometry.

99%+
Purity Target
HPLC
Purity Testing
MS
Mass Spectrometry
3rd Party
Lab Verified
Independent Third-Party Laboratory Testing
Each batch is submitted to a third-party laboratory for analytical testing. Testing verifies compound identity and purity using HPLC and Mass Spectrometry.
Purity Standards
- Target Purity: ≥ 99%
- Third-Party Batch Testing
- Lot Tracking and Documentation
- Controlled Storage and Handling
Analytical Testing Methods
High Performance Liquid Chromatography (HPLC)
HPLC is used to separate, identify, and quantify each component in a compound, verifying purity and composition.
Mass Spectrometry (MS)
Mass spectrometry verifies molecular weight and confirms compound identity.
Verification Includes
- Identity Verification
- Purity Confirmation
- Molecular Weight Verification
- Batch Consistency
- Third-Party Lab Reports
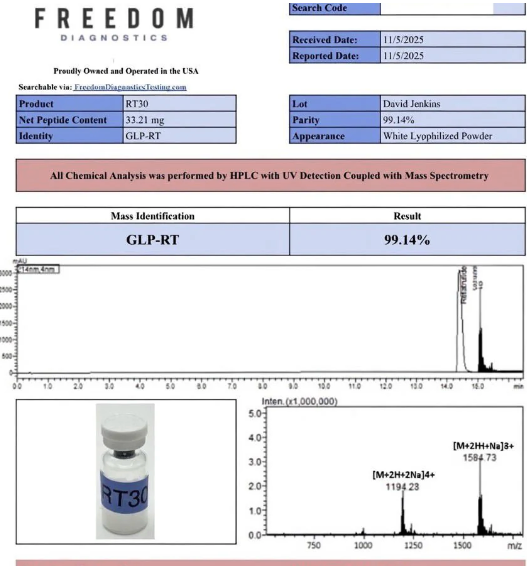
Certificate of Analysis (COA)
Each batch is tested by a third-party laboratory to verify identity, purity, and composition.
- Testing Laboratory: Freedom Diagnostics
- Testing Method: HPLC + Mass Spectrometry
- Verified Purity: 99.14%
- Appearance: White Lyophilized Powder
- Net Content Verified: 33.21 mg
Storage & Handling
Products are stored in controlled environments and handled according to laboratory handling procedures to maintain stability and integrity.
Research Use Only
All products are intended for laboratory research use only and are not for human consumption.
Understanding HPLC Purity Testing
High Performance Liquid Chromatography (HPLC) is the industry standard method for quantifying peptide purity. During HPLC analysis, a dissolved sample is passed through a column packed with a stationary phase. As the mobile phase carries compounds through the column at different rates, they separate into distinct peaks on a chromatogram. Each peak's area is proportional to the quantity of that component in the sample.
For a research peptide, the target compound should represent the dominant peak — ideally 99% or greater of the total peak area. Impurities appear as smaller adjacent peaks. A well-characterized HPLC report includes the retention time, peak area percentage, and a UV trace at 214–220 nm (the wavelength at which peptide bonds absorb).
Alpha Tides requires that each batch achieve ≥99% purity by HPLC before release. Batches that fall below this threshold are not distributed. This standard is higher than many generic chemical suppliers who release material at 95–98% purity.
Mass Spectrometry Identity Confirmation
HPLC purity alone does not confirm that the correct compound is present — only that most of the material is a single substance. Mass spectrometry (MS) provides identity confirmation by measuring the mass-to-charge ratio of ionized molecules.
Each peptide has a unique molecular formula and, consequently, a unique monoisotopic mass. The observed m/z values in MS analysis are compared against the theoretical mass for the target peptide. A match within acceptable tolerance (typically ±0.05 Da for high-resolution instruments) confirms the compound identity. For longer peptides where multiply charged ions are expected, the charge-state distribution pattern also confirms sequence integrity.
Together, HPLC and MS provide two independent data points that confirm both the purity and identity of the research material. This dual-verification approach is what distinguishes research-grade peptides from lower-grade synthesis outputs.
Lyophilization and Stability
All Alpha Tides peptides are supplied in lyophilized (freeze-dried) form. Lyophilization is a multi-stage process: the peptide solution is first frozen to below its eutectic point, then subjected to primary drying under vacuum (sublimation removes ice directly without a liquid phase), and finally secondary drying reduces residual moisture to below 1%.
The result is a dry, porous powder that retains the peptide's molecular structure with high fidelity. Lyophilized peptides are significantly more stable than solutions — stored at −20°C, properly lyophilized peptides maintain integrity for two or more years. In contrast, reconstituted peptide solutions may begin degrading within days to weeks, depending on the compound and storage conditions.
Researchers should store lyophilized vials at −20°C, away from light and moisture, until ready for reconstitution. Once reconstituted with bacteriostatic water, store at 2–8°C and use within 28–30 days. See the Reconstitution Protocol and Storage Protocol for detailed handling instructions.
What Researchers Should Ask Any Supplier
When evaluating a peptide supplier for research purposes, the following questions help distinguish reliable, research-grade sources from lower-quality alternatives:
- Is testing performed by an independent third-party lab? In-house testing has an inherent conflict of interest. Third-party testing removes that bias.
- Is a Certificate of Analysis (COA) available per batch? Generic COAs that don't reference a specific lot number are not meaningful quality documentation.
- What testing methods are used? HPLC and MS together are the minimum standard. Suppliers using only visual inspection or internal spectroscopy provide much weaker assurance.
- What is the purity specification? 99%+ by HPLC is research-grade. Lower specifications introduce higher uncertainty into experimental results.
- How are products stored and shipped? Lyophilized peptides should be shipped cold or with proper desiccant to prevent moisture ingress during transit.
Alpha Tides publishes batch COA documentation and uses Freedom Diagnostics as its third-party analytical laboratory. Lot-specific reports are available for active inventory.